
We have developed multilingual packaging files for a wide range of markets. In Europe, that has included packages featuring as many as seven languages, while in Canada we regularly produce bilingual English/French files. Each market brings its own challenges, and here we’ll look at a few different approaches.
In Canada, both languages must appear with equal prominence, including the same size and visual weight. However, the exact requirements depend on where the product will be sold. If the product is intended for Quebec, all packaging text must be translated into both English and French. If the product is being sold elsewhere in Canada but not in Quebec, only the product name, descriptor, contents declaration, and required legal text must be bilingual. Marketing copy does not need to be translated, which can make for a cleaner, less crowded package. That said, if a retailer in Quebec later requests the product, the package must be redesigned to meet Quebec standards. For that reason, we generally recommend translating to the stricter Quebec requirements from the outset.


In this example, the package is a window box with a relatively open front panel, giving us enough room to incorporate the additional bilingual text. One area that requires particular care is the contents declaration on the front. In the U.S., the minimum size of the numerals is determined by a formula based on the area of the principal display panel. Canada uses a similar approach, but the calculation differs. As a result, simply translating the U.S. package without adjusting the type size can often lead to non-compliance in Canada.


The images below show a different solution for bilingual packaging. This package originally featured three windows on the front panel. While it would have been possible to make that front bilingual, the result would have been overly crowded and difficult to read.


Instead, we recommended removing the marketing copy from the back panel, eliminating the front windows, and replacing them with product images. This allowed us to create two “front” panels, one in English and one in French. The ingredient component listing was moved to the side panels, while the UPC and legal text were placed on the bottom panel. This approach created a cleaner presentation while still meeting bilingual requirements.


Food packaging raises many of the same challenges as toy packaging when creating bilingual files. As shown in the images below, the English-only packaging and the bilingual Canadian packaging use different type sizes for the net contents statement. This difference reflects the separate regulatory formulas used in each country.



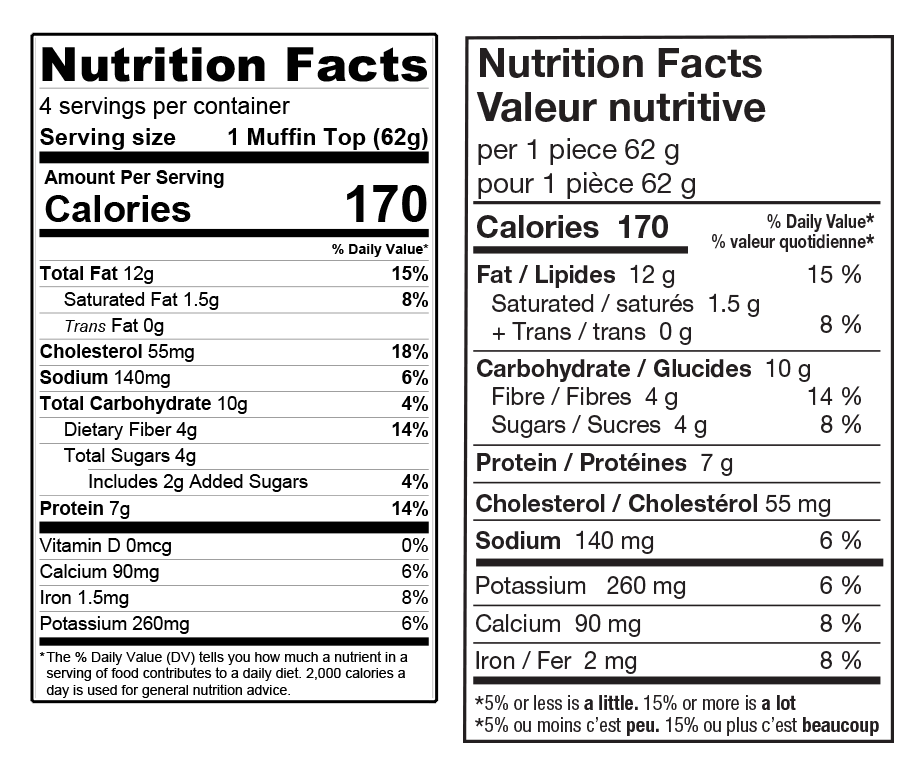
Nutrition Facts panels are another area where direct translation is not enough. In the U.S., these panels are regulated by the FDA, while in Canada they fall under the Canada Food Inspection Agency (CFIA). The Canadian panel is not simply a translated version of the U.S. format; the required information, structure, and sizing rules are different. In the U.S., the size of the Nutrition Facts panel is tied to the size of the principal display panel. In Canada, the CFIA determines panel size based on a percentage of the total available packaging surface, including the front, back, and sides.
Canada also now requires front-of-package labeling for products high in fat, sugar, and sodium, adding another important consideration for packaging intended for that market.